Medical Touchscreens Are Not an Accessory — They Are the Core of Device Stability
Factory-direct medical touchscreens with fully controlled quality, custom engineering support, and consistent batch performance — built for devices where downtime is not an option.
Request Medical Touchscreen SolutionKnow the Stakes
A Touch Failure in Medical Devices
Is Never Just a Repair Issue
When a touchscreen malfunctions in a medical environment, the consequences cascade far beyond the hardware itself. Understanding the full chain of risk is the first step to preventing it.
Device Downtime
A failed touchscreen can halt critical equipment mid-operation. In diagnostic or monitoring devices, every hour offline directly impacts patient care capacity and department workflow.
Clinical Operation Errors
Unresponsive or misregistered touch inputs introduce real risk during sensitive procedures. Operators face elevated cognitive load, increasing the likelihood of dangerous input mistakes.
Brand Trust Damage
In the medical market, quality incidents travel fast. A single reliability issue can undermine years of brand equity, trigger costly recalls, and permanently damage hospital relationships.
The Critical Difference
Why Standard Industrial Touchscreens
Cannot Serve Medical Devices
Industrial touchscreens are built for durability under mechanical stress. Medical touchscreens demand an entirely different set of guarantees — ones that no industrial spec sheet can substitute.
Stability Over Specs
Medical devices operate 24/7 in temperature-controlled environments where "good enough" performance is not acceptable. Industrial screens are typically rated for sporadic high-load use — the opposite of what a continuously active patient monitor or infusion pump demands. Medical-grade panels must maintain consistent brightness, touch sensitivity, and response uniformity across years of uninterrupted operation.
Consistency Across Production Batches
Industrial supply chains often allow component-level substitutions between batches — acceptable for factory automation, catastrophic for medical OEMs who go through regulatory validation. Any BOM change in a medical device triggers re-validation cycles. Your touch supplier must deliver exact component consistency, order after order, year after year.
Sample Approval ≠ Production Safety
Passing a sample approval is the beginning of a medical supplier's responsibility, not the end. The real test is whether the 500th unit performs identically to the first. Industrial suppliers often lack the process controls — same BOM enforcement, batch traceability, in-process QC — to guarantee that mass production stays aligned with the validated sample.



If your current supplier cannot answer these three questions, it's time to reconsider the source.
Speak with a Medical Touch SpecialistThe right touchscreen isn't the one that passes your sample review — it's the one that performs identically on unit 1,000.
Medical device manufacturers require suppliers who understand validation cycles, batch discipline, and long-term support — not just competitive pricing on a first order.
Where Our Touchscreens Work
Designed for the Medical Environments Where Precision
Cannot Be Compromised
Six Applications. One Standard: Zero Failure Tolerance.
Each medical application places unique demands on a touchscreen interface. Here's where our solutions perform — and what failure looks like in each context.
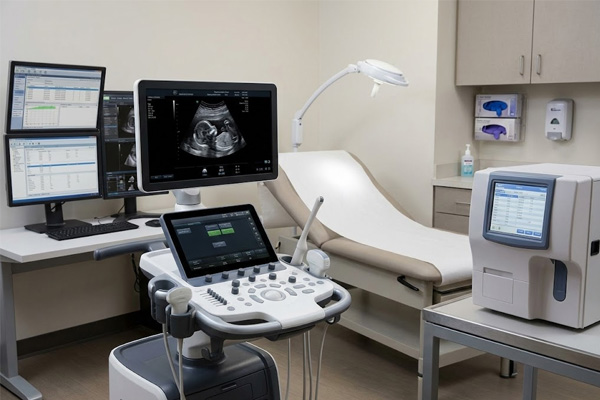 Diagnostic Equipment
Diagnostic Equipment
Diagnostic Equipment
In imaging systems, ultrasound consoles, and laboratory analyzers, touchscreen interfaces manage real-time parameter input. Displays must maintain calibrated sensitivity across long shifts and resist interference from nearby electromagnetic sources without any degradation in positional accuracy.
Key risk: Touch drift causing input offset in precision settings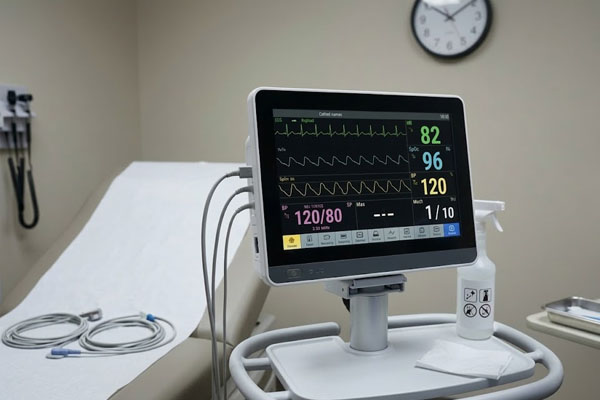 Patient Monitoring
Patient Monitoring
Patient Monitoring Systems
Bedside monitors operate around the clock, often in environments with frequent cleaning using alcohol-based agents. Touchscreens must maintain chemical resistance without surface degradation while providing reliable response to both gloved and bare-finger touch at all hours.
Key risk: Surface delamination from repeated disinfection cycles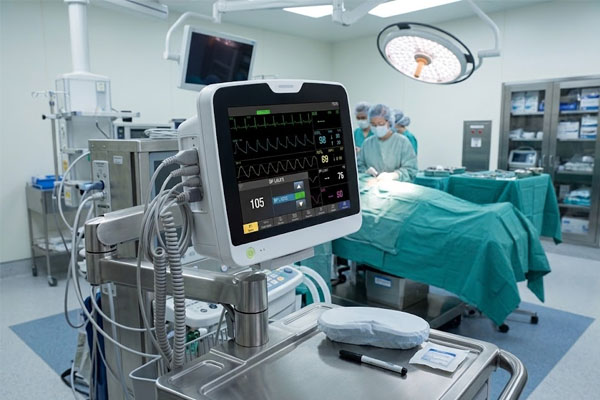 Medical Control Panels
Medical Control Panels
Medical Control Panels
Surgical tables, anesthesia delivery systems, and infusion pump arrays rely on integrated touch panels for real-time parameter control. In these environments, a single unresponsive touch input can interrupt an active procedure — making screen reliability a patient safety issue, not just a UX concern.
Key risk: Touch dropout during active surgical use Portable Devices
Portable Devices
Portable Medical Devices
Point-of-care devices, handheld diagnostic tools, and mobile clinical workstations require slim, lightweight touchscreens with low power draw and shock resistance. Customization in size, thickness, and connection interface is typically non-negotiable for OEM integration.
Key risk: Off-spec dimensions causing integration failure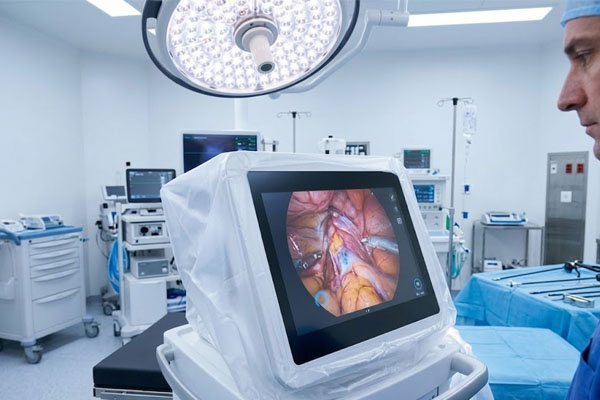 Surgical Imaging
Surgical Imaging
Surgical Imaging Systems
Intraoperative imaging displays and laparoscopic visualization systems demand touchscreens that perform flawlessly under sterile drape conditions, high-intensity lighting, and gloved multi-touch interaction — simultaneously. Any input lag or calibration drift in the OR is not a usability issue; it is a clinical risk event.
Key risk: Calibration drift under sterile drape conditions Rehabilitation Equipment
Rehabilitation Equipment
Rehabilitation Equipment
Physical therapy devices, biofeedback systems, and motor training platforms require highly responsive touchscreens that register light, imprecise touch inputs from patients with reduced motor control. Surface durability under constant physical contact and institutional cleaning schedules is a primary performance requirement.
Key risk: Sensitivity loss under repeated light-touch patient useTell us your device category and integration requirements — we'll provide a tailored solution brief within 24 hours.
Request Application-Specific ConsultationOur Medical Solutions
From Component to Integrated System —
Solutions Built Around Your Device Requirements
We don't sell off-the-shelf screens. We provide engineered touchscreen solutions that are sized, specified, and validated to match your exact device design — with supply continuity guaranteed.
Medical Touch Displays
Standalone medical-grade touchscreen displays with configurable brightness, anti-glare coating, and hardened surface treatment for clinical environments. Available from 5" to 21".
- High-brightness option for OR lighting
- Chemical-resistant surface coating
- IEC 60601-1 compatible design
- Custom aspect ratios and bezels
Touch Panel + Display Integration
Fully bonded assemblies that eliminate air gaps, reducing condensation risk and improving optical clarity. Ideal for handheld and portable devices where space is constrained.
- Optical bonding for clarity and durability
- Reduced profile for compact enclosures
- Glove-compatible PCAP or resistive
- Full drawing-based customization
Long-Life Medical Touch Modules
Designed for 7–10 year lifecycles. Comes with extended component availability commitments, frozen BOM documentation, and scheduled production windows.
- 5+ year supply continuity commitment
- BOM freeze and change notification
- Batch traceability documentation
- Lifecycle management support
OEM Engineering Support
Medical Device Customization Is Not Optional —
It's Where a Real Factory Proves Its Value
Generic catalogue screens rarely fit the precise enclosure, optical, and certification requirements of medical OEM devices. Here are the five customization dimensions we address at the engineering stage — before a single production unit is committed.



Size & Structural Dimensions
Custom screen dimensions — including non-standard aspect ratios, cutout shapes, and mounting hole layouts — are produced directly from the customer's mechanical drawing. Our engineers confirm all critical tolerances before tooling begins to eliminate misfit at the assembly stage.
Drawing-confirmed before toolingGlass Thickness & Surface Treatment
Glass specification — including cover lens thickness, chemical strengthening level, anti-glare or anti-fingerprint coating, and edge finishing — is defined and frozen during the sample stage. Medical devices that undergo regular chemical disinfection require surface specs that survive years of protocol without degradation.
Hardening spec fixed at sample freezeTouch Technology Selection
We support PCAP (projected capacitive), resistive, and infrared touch technologies — each with specific advantages for gloved use, fluid resistance, and precision input. The right choice depends on your intended environment, user workflow, and sterilization protocol. We guide the selection before engineering commits.
Protocol-matched touch technologyBrightness, Backlight Life & Calibration
Operating room and high-ambient-light environments demand displays rated above 700 nits. Long-life medical applications require backlight modules with verified 50,000+ hour ratings and consistent luminance output across the operational lifecycle. We specify and document these parameters at the component level.
Backlight spec documented and traceableMounting Interface & Installation Method
Panel-mount, VESA, recessed chassis, and custom bracket configurations are all supported. Interface connectors — signal, power, and touch controller — are confirmed against your system architecture during the engineering review, eliminating integration surprises during final assembly.
Connector map confirmed before productionRegulatory Documentation Support
We provide full engineering documentation packages — including material declarations, component datasheets, test reports, and change notification agreements — structured to support your IEC 60601, FDA 510(k), or CE submission requirements without additional back-and-forth.
Documentation ready for submissionSend us your device drawings or specification brief — our engineers will return a customization feasibility review within 2 business days.
Submit Your Customization RequirementsEngineering Process
How We Prevent "Understanding Gaps"
from Drawing to First Production Run
Most quality problems in custom medical touchscreens don't begin on the production line — they begin in a miscommunicated specification. Our three-stage engineering confirmation process closes these gaps before they cost you a re-validation cycle.
Drawing-Based Confirmation
Every project begins with a structured review of your mechanical and optical drawings. Our engineers annotate critical dimensions, flag potential tolerance conflicts, and issue a written confirmation document — not a verbal approval — before any engineering work begins. This creates a shared reference point that both teams can return to at every stage.
Output: Signed drawing confirmationEngineering Confirmation Review
Once the sample is built, our engineering team conducts a systematic comparison against the confirmed drawing — checking all critical dimensions, functional performance, and interface compatibility. Any deviation triggers a documented change request, not a silent correction. The customer sees every discrepancy and approves every resolution before the sample is approved for next-stage use.
Output: Engineering deviation logSample Freeze Mechanism
Before mass production begins, the approved sample is formally frozen: a physical sample is archived, the full BOM is documented and locked, and the production engineering file is closed. Any future change — however minor — requires a new engineering review cycle. This mechanism ensures the 1,000th unit is built from the same specification as the validated sample.
Output: Frozen sample + locked BOM fileWhy this matters for medical OEMs: Regulatory validation is tied to a specific product configuration. Any undocumented component change after validation approval creates compliance risk. Our sample freeze mechanism is designed specifically to protect the integrity of your validated configuration through the full production lifecycle.
Ready to start your engineering review? Share your drawing files and we'll open a project confirmation process immediately.
Start Engineering ConfirmationProduction Risk Management
Sample Approval Is the Beginning —
Mass Production Is Where Most Suppliers Fail
Most medical device engineers know to scrutinize a sample. Far fewer focus equally on what happens between the sample approval and the 500th production unit. This is where undisclosed component substitutions, process drift, and batch inconsistency introduce the real risk.
Same BOM — No Silent Substitutions
Every production batch is built from the exact component list that was approved at sample freeze. We maintain a locked BOM database and require a documented engineering review for any proposed change — including equivalent substitutions. If a component becomes unavailable, customers are notified before production, not after delivery. Your validated configuration stays intact.
Enforced BOM lock from sample through EOLBatch Consistency Controls
Each production batch goes through a structured first-article inspection that compares a randomly selected unit against the frozen sample's critical parameters. Dimensional, optical, and touch performance results are recorded against defined tolerance bands. Batches that fall outside limits are quarantined and reviewed — never shipped with a note attached.
First-article inspection on every production batchFull Inspection Records & Traceability
Inspection records are maintained for every production batch — including incoming material verification, in-process measurement data, and final test results. Each shipping lot is assigned a traceable batch code that links back to the production run records. If a quality question arises six months after delivery, we can pull the production data the same day.
Full lot traceability from incoming material to shipment
Ask us how we handle BOM changes and batch traceability — the answers will tell you everything you need to know about a supplier's production discipline.
Request Our Production Control OverviewQuality System
A Quality Control System Built
Around What Medical Customers Actually Audit
Medical device procurement teams look beyond product specs. They audit process discipline — incoming material controls, in-process monitoring, final verification, and traceability infrastructure. Here is how our QC system maps to each of these expectations.
Incoming Inspection
All incoming display panels, touch sensors, cover glass, and electronic components are subject to a structured IQC process before entering production inventory. Components that fail specification — including those from approved suppliers — are quarantined, documented, and returned, never reworked into the production flow without formal disposition approval.
- Material certificate verification against PO spec
- Dimensional sampling per AQL level
- Optical and functional spot-check testing
- Quarantine tagging with disposition record
In-Process Control
Critical assembly stages are governed by defined inspection checkpoints — not end-of-line discovery. Bonding layer uniformity, touch controller calibration, and connector seating are verified at the assembly station before the unit moves forward. Process deviations are logged immediately and reviewed by QC supervision before the affected units continue.
- Station-level inspection checkpoints at critical joins
- Bonding uniformity measurement during assembly
- Touch controller calibration verification in-line
- Real-time process deviation logging
Final Inspection
Every finished unit undergoes a 100% final inspection against a defined acceptance criteria checklist before packaging. Cosmetic, functional, dimensional, and electrical parameters are verified. The inspection result is recorded on the unit's production record. Units that fail final inspection are clearly segregated — batch shipping is halted until disposition is complete.
- 100% functional power-on test per unit
- Touch accuracy and linearity verification
- Cosmetic grading against approved sample
- Dimensional final-check on critical interfaces
Traceability System
Each shipping lot is assigned a unique batch code that maps to: the specific production run records, the incoming material certificates used, the in-process inspection data, and the final inspection results. In the event of a field quality question, we can provide the full production record for any delivered unit within 24 hours — regardless of when it was shipped.
- Unique batch code on every shipping carton
- Material certificate archive per production run
- Inspection data linked to batch traceability
- Record retention beyond standard product lifecycle
Production Quality Gate Sequence
Inspection
Checkpoints
Calibration
Inspection
Assignment
Record Lock
We welcome supplier audits. Request our QC documentation package or schedule a factory audit visit.
Request QC Documentation PackageDirect from the Source
We Manufacture — We Don't Resell.
And That Difference Is Critical for Medical OEMs.
Trading companies can quote fast and pivot faster — but they cannot commit to frozen BOMs, engineering reviews, audit access, or supply continuity, because they don't control production. With Yinghong, you are speaking to the source.



Direct Factory Access
Customers communicate directly with the engineering team that builds the product. There is no intermediary layer translating your requirements — specifications travel without distortion from your desk to the production line. When you request a change, an audit, or a production record, you get a direct answer from the people who hold the information.
Medical Project Track Record
Our medical touchscreen experience covers diagnostic displays, portable patient monitoring units, point-of-care terminals, and surgical control interfaces — across customers in Europe, North America, and Asia-Pacific. We understand the validation cycle, the documentation requirements, and the supply continuity expectations that come with each type of medical device project.
Export-Oriented Manufacturing
Our production is structured for international OEM supply. Export documentation, CE and UL component certifications, multi-language technical files, and structured communication for overseas procurement teams are standard deliverables — not special arrangements. Our customers in Europe and North America work with us because the process feels like a local supplier with factory-direct pricing.
Verify the source yourself — request a factory introduction, audit documentation, or schedule a video walkthrough of our facility.
Request Factory Verification PackageLong-Term Partnership Value
Why Medical Device Companies Return —
Year After Year, Project After Project
The decision to switch touchscreen suppliers mid-lifecycle carries enormous validation and compliance costs. Medical OEMs choose long-term partners for specific, structural reasons — not just because the relationship is comfortable.
Stable Supply Without Re-Validation
Once a component is validated in a medical device, every supplier change triggers a new qualification cycle — a process that can take months and carry significant regulatory cost. Long-term customers stay because we guarantee supply continuity with frozen BOM documentation. No surprise substitutions. No re-validation surprises. The product you validated is the product that ships — for the full lifecycle of your device.
Fast Technical Response When It Matters
When a field quality question surfaces — an unexpected touch behavior, a cosmetic deviation in a late batch, a connector compatibility question during a new product revision — response time matters more than it ever did at the quotation stage. Long-term customers value that they reach our engineering team directly, not a customer service queue, and receive a technical answer the same day, not three business days later.
Predictable Risk Across Product Generations
Medical OEM product cycles are long, and device families often share common touchscreen interfaces across generations. Long-term suppliers who understand the full device context — and whose production team knows the product history — deliver significantly lower integration risk when a new revision or next-generation product is developed. Knowledge continuity has real engineering value that a new supplier quote cannot include in their price.
Value Measured in Risk Reduction, Not Just Price
The lowest price in a first-order negotiation is rarely the lowest total cost of medical device supply. Hidden costs — undisclosed component changes that trigger re-qualification, quality incidents that delay shipments, slow technical responses that extend a field issue investigation — accumulate quickly. Long-term customers stay because they have already measured what supply stability and process discipline actually save across a multi-year production program.
Long-term supply relationships start with the right first conversation. Tell us your device program and timeline.
Discuss a Long-Term Supply ProgramHow We Work Together
From First Requirement to Long-Term Supply:
A Process Designed to Protect Your Program
Every step of our cooperation process is structured to reduce your decision risk — not to accelerate our sales cycle. Here is exactly what working with Yinghong looks like from inquiry to repeat order.
Requirement Review
We review your device application, volume profile, regulatory environment, and key technical constraints. This is an engineering conversation, not a sales call — the outcome is a clear shared understanding of what the touchscreen must deliver, not just what it must look like.
Output: Requirement alignment documentTechnical Confirmation
Our engineering team reviews your mechanical drawings, interface specifications, and performance requirements. All critical dimensions and functional parameters are confirmed in writing before any production engineering begins — preventing misunderstandings from becoming hardware.
Output: Signed technical confirmationSampling
Engineering samples are produced against the confirmed drawings. Sample delivery includes a dimensional inspection report and functional test data. Customers receive the information to make an informed approval decision — not just a physical unit to evaluate by feel.
Output: Samples + inspection reportValidation Support
We provide the technical documentation your validation team needs: component certificates, material specifications, dimensional data, and test records. If your validation protocol requires additional testing data or engineering clarification, our team is available throughout the process — not just at the delivery stage.
Output: Full validation documentation packageMass Production
Production runs against the frozen BOM and locked sample specification. Every batch undergoes incoming inspection, in-process checkpoints, and 100% final testing before shipping. Batch records and traceability documents are available upon request for every delivered lot.
Output: Production + full batch recordsRepeat Orders
Repeat orders execute against the original frozen specification — no re-engineering, no re-confirmation, no cost of re-establishing the technical baseline. We maintain production readiness for all active customer programs. Supply lead times for repeat orders are significantly shorter than for new development projects.
Output: Consistent supply across device lifecycleThe real value of this process: from the second order onward, you are not re-spending the qualification investment. The technical foundation is already in place. Each repeat order draws directly from the validated specification — delivering consistent product faster, with lower total procurement overhead across the device's commercial life.
Start the process with a requirements conversation — no commitment required at this stage.
Begin Your Requirement ReviewVerified Customer Feedback
What Medical Device Teams Say After
Working with Yinghong
Long-term supplier relationships are built on real performance — not first-order impressions. These are the reasons our medical customers continue to expand cooperation over multiple programs and years.
We moved three SKUs over to Yinghong after our previous supplier introduced a backlight substitution that triggered a full re-qualification cycle — without notifying us in advance. Since the switch, we have run four production batches with zero BOM deviations. Their engineering team responded within the same business day every time we raised a technical question. That responsiveness has real value when you are managing a regulated supply chain.
The validation documentation package Yinghong provided was complete on the first submission — no back-and-forth requesting missing certificates or additional test data. That alone saved us two weeks in our IQ/OQ cycle. The engineering confirmation process also caught a connector interference issue at the drawing stage that would have caused a board redesign if it had reached hardware. This is the kind of supplier capability that pays for itself before the first production order ships.
We source touchscreens for three product lines across two generations. Yinghong manages all three programs with consistent quality and without confusion between product configurations. When we started development on our Gen 2 platform, the engineering team already understood our enclosure constraints and regulatory context — we did not need to re-educate a new supplier. That accumulated knowledge has concrete value when you are under a product launch timeline.
Request reference details or a customer introduction for your device category.
Request Customer ReferenceCommon Questions
Questions Medical Procurement Teams
Ask Before Approving a New Touch Supplier
We understand the due diligence process. Here are the questions we hear most often — answered directly.
Factory Identity Are you a real manufacturer, or a trading company?
We are a direct manufacturer. Our production facility operates in Dongguan, China, with in-house assembly lines, a dedicated engineering team, and on-site QC infrastructure. We do not outsource production to third-party factories or act as an intermediary between customers and other manufacturers.
We welcome factory audit requests — virtual walkthroughs and on-site audit visits can be arranged for qualified customers. We can provide business registration documents, facility photographs, and production capability data prior to a formal audit visit.
Custom Engineering Can you support medical-grade customization, including non-standard sizes and surface treatments?
Yes. Custom engineering is a core capability, not a special arrangement. We support:
- Non-standard dimensions from customer-supplied mechanical drawings
- Custom glass thickness, chemical strengthening level, and surface treatments (anti-glare, anti-fingerprint, oleophobic coating)
- PCAP, resistive, and infrared touch technology — selected based on your clinical use environment
- High-brightness backlight specifications for OR and high-ambient-light applications
- Custom mounting interfaces, connector types, and cable routing
All customization begins with a drawing review and written engineering confirmation before production tooling is committed.
Batch Consistency How do you ensure batch consistency across repeat orders?
Batch consistency is maintained through three mechanisms:
- Locked BOM: The component list is frozen at sample approval. Any proposed change — including equivalent substitutions — requires a formal engineering review and customer notification before implementation.
- First-article inspection per batch: Each production run begins with a first-article comparison against the frozen sample's critical parameters. Batches that fall outside tolerance are quarantined before shipping.
- Batch traceability: Every shipped lot carries a batch code that maps back to the full production record — including incoming material certificates, in-process inspection data, and final test results.
We can provide a sample batch record package upon request so you can review the documentation structure before placing a production order.
Pilot Orders Do you support pilot or initial validation orders before committing to full production volumes?
Yes. We understand that medical device procurement involves a sequential qualification process before full production volumes can be committed. Our project structure accommodates this clearly:
- Engineering samples: Small quantities produced from confirmed drawings for mechanical fit, functional validation, and regulatory submission support.
- Pilot production orders: Limited production runs (typically 50–500 units) built under full production conditions — same process controls, same BOM, same inspection protocol as a standard production order. Pilot runs are used for IQ/OQ/PQ validation and clinical trial supply.
- Commercial production: Full volume production against the validated specification following pilot completion.
Pilot orders are priced differently from production orders, but they are built with exactly the same process discipline — because they feed directly into your validation record.
Export Markets Which markets and regions do you currently export to?
Our medical touchscreen products are currently supplied to OEM manufacturers in Europe (primarily Germany, Netherlands, and UK), North America (United States and Canada), and Asia-Pacific (Japan, South Korea, and Australia).
Our export infrastructure — including CE-compatible component certifications, English-language technical documentation, and structured international logistics — is built for international OEM supply as a standard operation, not a special arrangement. We are familiar with the documentation requirements for CE-marked medical devices and FDA-registered device supply chains, and can discuss specific certification documentation requirements for your target market during the project review stage.
Have a specific question not covered here? Our engineering team responds within one business day.
Send Us Your Technical QuestionGet Started Today
Medical Touchscreens Are About Risk Control.
Not Just Price.
Every day a medical device runs in the field with an unvalidated or inconsistently supplied touchscreen is a risk your QA team is carrying silently. The right source eliminates that risk — for the full lifecycle of your device, not just the first batch.
Tell us your device, volume, and timeline — we'll respond within 1 business day.Request Your Medical Touchscreen Solution
Complete the fields below — the more context you provide, the more specific our response will be.
